Volatile organic compounds (VOCs) are most commonly emitted from oil refining and oil & gas extraction sites as well as power generation and food and beverage facilities.
VOC emissions pose a double-edged threat: Not only can VOC compounds cause nuisance complaints from nearby residential areas encroaching on industrial sites, they’re also heavily-regulated pollutants known to harm people, wildlife and the environment.
Accordingly, the U.S. Environmental Protection Agency has worked alongside industry leaders to develop VOC testing methods that identify and quantify emissions. The emission assessments we provide are a critical early step toward developing a solution that reduces or eliminates these harmful emissions.
VOC testing methods TO-15, TO-17 and ASTM D5504
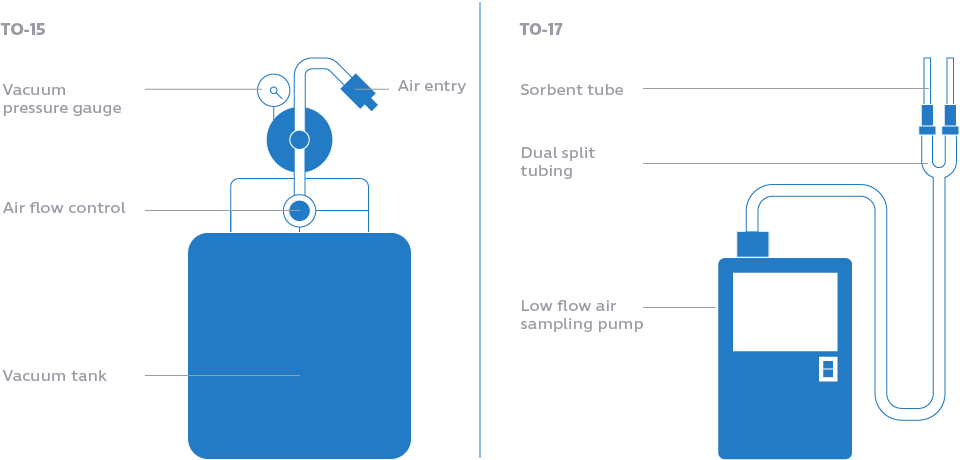
The EPA’s Method TO-15 outlines proper VOC testing by collecting air in a specially-prepared stainless steel vacuum canister. Air is drawn into the canister through a flow controller for as little as an hour or as long as two days. Then, the canister is sent for laboratory analysis. Collection of air samples using VOC testing method TO-15 is shown in this video.
Method TO-17 differs in that air samples are collected through sorbent tubes instead of canisters. The sorbent traps VOCs as air passes through the tube. Prior to analysis, the sample is freed from the sorbent via thermal desorption. A TO-17 test kit is shown in this video.
Reduced organic sulfur compounds (ROSCs) commonly accompany volatile hydrocarbons as emissions from oil & gas processing facilities. Unfortunately, most conventional VOC testing methods don’t accurately detect sulfur compounds. In response, the ASTM D5504 testing method provides for the collection of air samples in inert plastic bags. Then, those samples are analyzed by sulfur chemiluminescence detectors (SCDs). Sulfur chemiluminescence detection is described in a later section.
Regardless of the VOC testing method used, the next step is analysis of the sample.
Gas chromatography and mass spectrometry (GC/MS)
Gas chromatography is a physiochemical separation technique, while mass spectrometry is an analytical method to analyze compounds by ionization. Each provides complementary information during VOC testing. To get the most comprehensive profile of the VOCs emitted from a site, the two methods are coupled together.
There’s rarely only one compound in an air sample. Usually, several VOCs are emitted from a facility. A gas chromatograph separates compounds in an air sample so that individual identification of compounds can be made by analytical detectors such as mass spectrometers (MS) or sulfur chemiluminescence detectors (SCDs).
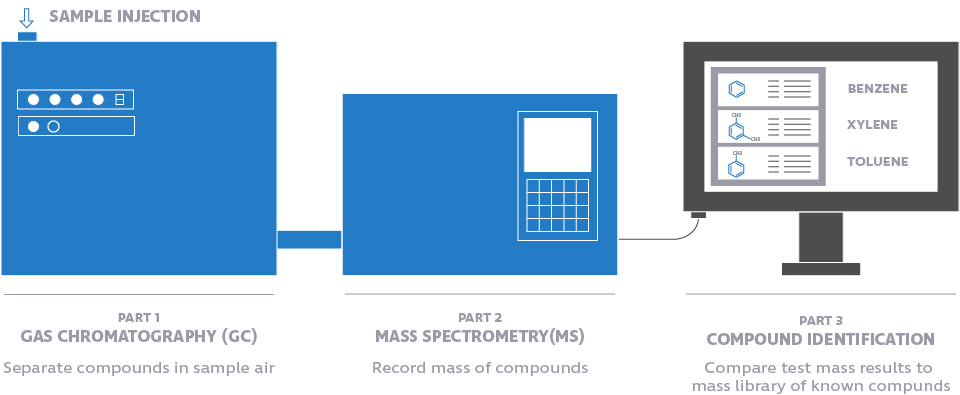
A mass spectrometer sends ionized atoms from the collected compounds through a sensitive filter that records their amounts and masses. Scientists then compare the data gathered from the mass spectrometer against a list of known compounds based on their mass, identifying both VOCs and ROSCs as well as determining their concentration. Watch this video to better visualize how GC/MS works.
Gas chromatography and sulfur chemiluminescence detection (GC/SCD)
Sulfur chemiluminescence is a detection method that quantifies sulfur concentration by recording the amount of light given off during a chemical reaction. Like mass spectrometry, sulfur chemiluminescence detectors are commonly coupled to gas chromatographs and analyze emission streams containing ROSCs through a two-stage process.
The first stage is the introduction of a sulfur-containing sample plus hydrogen (H2) and ambient air into a reaction furnace under vacuum to generate sulfur monoxide (SO). In the second stage, the sulfur monoxide is carried to a reaction chamber where it reacts with supplied ozone (O3). The result of that reaction is sulfur dioxide (SO2) and UV light.
The UV light generated by this reaction is directly proportional to the amount of sulfur in the sample. The light is measured by a photomultiplier tube; that value is then compared against known values.
Crafting your VOC emission solution
VOC testing and analysis tells us which compounds are emitted at a facility as well as each compound’s concentration. The next step is to run internal and governmental comparisons. The initial comparison is the analysis of VOC testing at your site against our database from previous jobs. It’s a quick way for us to determine the treatability of your challenge. If our database yields no results, our scientists will conduct a thorough literature review to see if your problem can be treated.
The other comparison is the sample VOC concentrations against statutory VOC limits. This is a critical step because not all VOCs are regulated equally. Comparing site sample data to statutory VOC limits will help us further craft the emission treatment solution perfectly tailored to the challenge at your site.
In the U.S., we comply with the EPA. But as an international company we’re well-versed in navigating the global patchwork of environmental regulations.
VOC testing is a key step to solving an industrial emission challenge. Our emission control experts take a meticulous approach to testing and analysis because that’s the foundation on which we create optimal treatment solutions. See how VOC testing fits into the rest of our process by reading up on BioAir Complete®.
Read our in-depth guide for more information about the sources of oil and gas emissions and methods for eliminating them. If you want to discuss an ongoing challenge at your facility or request a site audit, contact us now.
